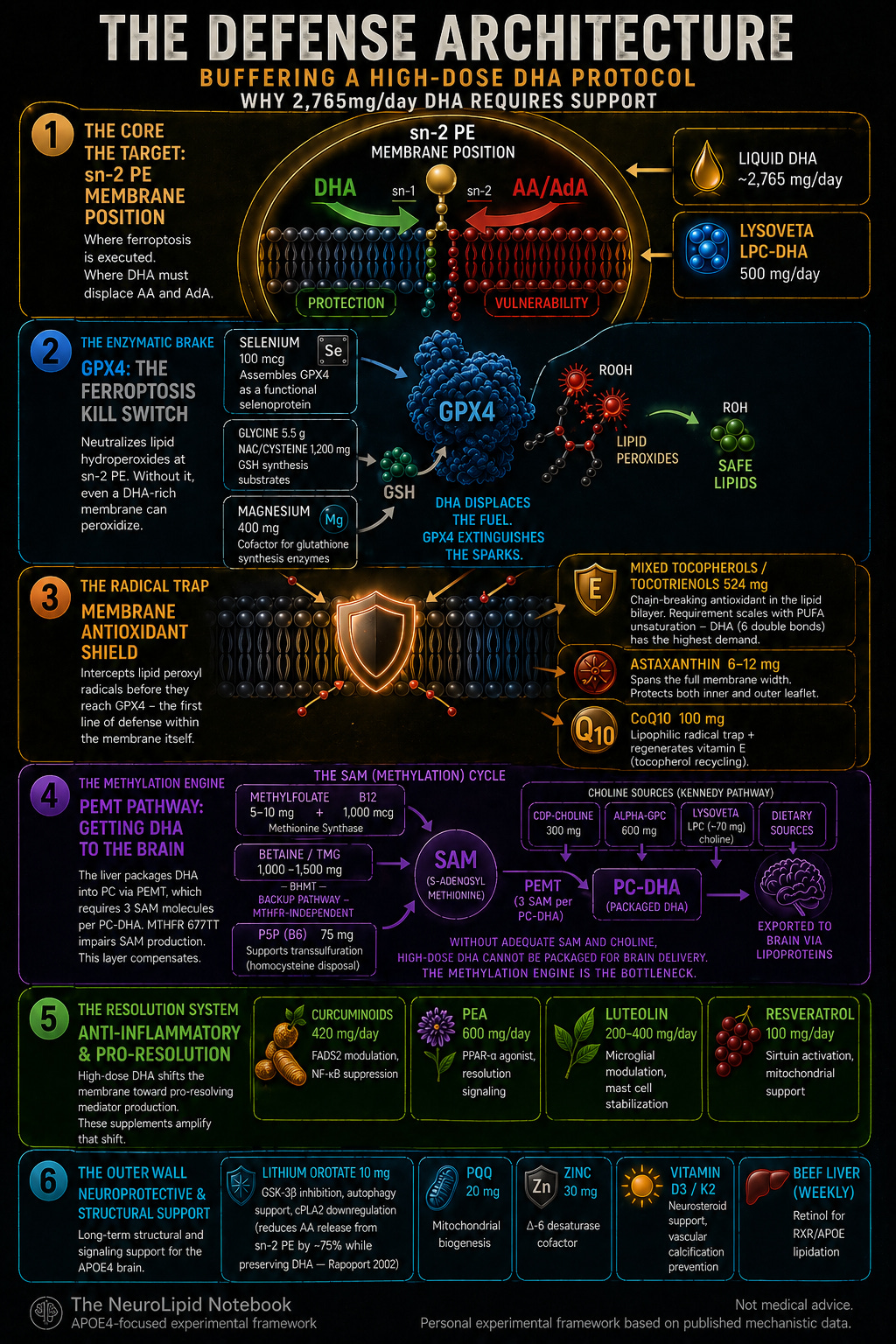
The Defense Architecture: Why 2,765 mg/day DHA Requires Support
Buffering my High-Dose DHA Protocol
Note: 6. Lithium at therapeutic doses reduces AA release from sn-2 PE by ~75% while preserving DHA
The Defense Architecture: Why 2,765 mg/day DHA Requires Support
If there is one idea to take from this piece, it’s this:
DHA is the most protective fatty acid in the brain—and one of the most vulnerable to oxidation.
At high doses, those two properties are inseparable.
This article explains the Defense Architecture—a layered framework built to ensure the first property wins. This is not “just” a supplement stack.
It is a system, where each component addresses a specific vulnerability created—or exposed—by high-dose DHA.
Why This Framework Exists: The Guidance Gap
While many of my research-heros have suggested that significant DHA is required for optimal health conditions in ApoE4 carriers. There is no clinical guidance for how to safely implement high-dose DHA supplementation. This is not an exaggeration. It is a factual description of the literature.
The European Food Safety Authority’s 2026 updated opinion retained a 1 g/day safe level for supplemental DHA alone, explicitly noting that “no UL for supplemental DHA alone can be established for any population group” due to insufficient dose-response data (EFSA, 2026). The 2012 EFSA opinion set a combined EPA+DHA ceiling of 5 g/day based on the absence of adverse effects at that level—not on evidence of safety at higher doses (EFSA, 2012). Neither opinion addresses antioxidant co-requirements, methylation demands, or genotype stratification.
The largest meta-analysis of omega-3 adverse effects (Chang et al., 2023; 90 RCTs) found no serious adverse events but did not assess oxidative stress management. The JACC review of omega-3 supplementation lists common adverse effects—GI symptoms, LDL elevation, atrial fibrillation risk at ≥2 g/day—without any mention of antioxidant buffering (Weinberg et al., 2021).
The antioxidant co-supplementation question is particularly barren. Sepidarkish et al. (2020) meta-analyzed 9 RCTs of omega-3 plus vitamin E and found that the combination significantly improved total antioxidant capacity and reduced malondialdehyde—a lipid peroxidation marker—compared to placebo. But no study has examined this in the context of APOE4 genotype, MTHFR variants, or brain-specific endpoints.
Perhaps most telling: Wander Du (2000) gave postmenopausal women high-dose fish oil (2.5 g EPA + 1.8 g DHA daily) with varying doses of vitamin E and found that fish oil increased plasma TBARS but adding vitamin E did not significantly reduce it. This study is frequently cited as evidence against co-supplementation. But it measured only plasma TBARS and oxidized protein—not membrane-specific peroxidation, not PE-specific hydroperoxides, and not in any genotype-stratified manner. The conclusion may be valid for cardiovascular endpoints in the general population. It says nothing about neuronal membrane ferroptosis vulnerability in APOE4 carriers.
The gap is real and specific: no guideline, consensus statement, or systematic review addresses how to manage the oxidative, methylation, and membrane turnover consequences of high-dose DHA in the context of APOE4 genotype, MTHFR variants, or brain-specific endpoints.
The Defense Architecture is a response to this absence—not an alternative to clinical guidance, but an attempt to build a framework where none exists, using the mechanistic science that does.
This post does not constitute individualized medical advice. The supplement framework described here reflects one person’s decisions for her own health, based on her interpretation of published mechanistic research. It has not been validated in clinical trials and should not be replicated without consultation with a qualified medical provider. Supplement interactions, contraindications, and individual risk factors vary — what is rational for one person’s genotype, methylation status, and clinical context may not be appropriate for another.
Nurse Jeannie is sharing her own opinions and her own decisions as an APOE4 carrier on a quest to make the best health decisions possible — for her.
Keep reading with a 7-day free trial
Subscribe to NeuroLipid Notebook to keep reading this post and get 7 days of free access to the full post archives.