Lithium Deficiency: A Hidden Early Factor in Alzheimer’s — What APOE4 Carriers Need to Know
An Evidence-Based Review
For decades, lithium has been known primarily as a psychiatric medication for mood stabilization. However, groundbreaking 2025 research by Aron et al. (available here: https://pmc.ncbi.nlm.nih.gov/articles/PMC12443616/ ) has fundamentally shifted our understanding of this element, revealing that endogenous lithium is dynamically regulated in the brain and plays a critical role in cognitive preservation during aging.
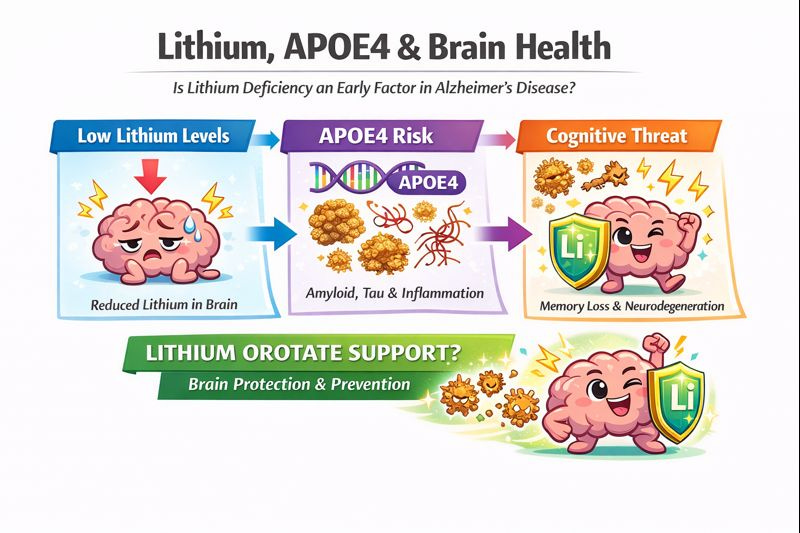
Lithium Deficiency as an Early Event in Alzheimer’s Disease
In individuals with mild cognitive impairment (MCI)—often the earliest detectable stage of Alzheimer’s disease—lithium was the only metal significantly reduced in brain tissue. As AD progresses, lithium bioavailability decreases further, partly because amyloid-β sequesters it ( https://pubmed.ncbi.nlm.nih.gov/40770094/ ).
Experimental studies in mice confirmed that reducing cortical lithium by approximately 50% accelerated classical AD pathology: amyloid-β deposition, tau phosphorylation, pro-inflammatory microglial activation, synapse and myelin loss, and cognitive decline—all mediated, at least in part, through GSK3β kinase activation. Single-nucleus RNA sequencing revealed that lithium deficiency produces transcriptome changes in multiple brain cell types that overlap with changes seen in AD.
Importantly, lithium orotate prevented these pathological changes and preserved memory in both AD-model and aging wild-type mice. While lithium orotate showed promise in this preclinical study, it should be noted that the evidence for differential pharmacokinetics between lithium salts remains mixed, with older studies from the 1970s finding no differences between lithium orotate and lithium carbonate, though more recent research suggests potential advantages in mouse models.
Clinical Evidence for Lithium in Cognitive Decline
Beyond preclinical findings, clinical trials have demonstrated benefits of low-dose lithium in MCI and AD. A landmark 2019 randomized controlled trial by Forlenza et al. showed that lithium carbonate at subtherapeutic concentrations (0.25-0.5 mEq/L) attenuated cognitive and functional decline in amnestic MCI over 2 years and increased CSF amyloid-β₁₋₄₂. Meta-analyses confirm that lithium therapy reduces dementia and AD risk. Read his study here: https://pubmed.ncbi.nlm.nih.gov/30947755/
The APOE4 Connection: Theoretical Implications
The APOE ε4 allele remains the strongest genetic risk factor for late-onset AD, with approximately 60% of clinically diagnosed AD patients carrying at least one APOE4 allele. Oc course, if you’ve found my Substack, you probably already know this and are fighting the good fight alongside me and other positive carriers.
APOE4 carriers experience earlier amyloid-β accumulation, accelerated disease onset, and more severe pathology compared to carriers of the protective ε2 or neutral ε3 alleles.
The Aron et al. study did not stratify findings by APOE genotype, and to date, no research has examined whether APOE4 carriers are differentially affected by lithium deficiency or respond differently to lithium supplementation.[1] However, several theoretical considerations suggest APOE4 carriers might be uniquely vulnerable to lithium depletion:
Convergent amyloid pathways: APOE4 impairs amyloid-β clearance and promotes earlier, more abundant amyloid deposition. Since lithium deficiency promotes amyloid-β accumulation via GSK3β activation, APOE4 carriers could experience a synergistic effect, with both impaired clearance mechanisms and reduced lithium-mediated neuroprotection simultaneously driving pathology.
Lipid metabolism disruption: APOE4 has relatively lower lipid-binding efficiency compared to APOE3, impairing cholesterol efflux and leading to lipid droplet accumulation and lysosomal dysfunction in neurons and glial cells. APOE4 also disrupts the LXRβ/ABCA1 pathway in oligodendrocytes, causing sphingolipid metabolism imbalance and demyelination. Lithium’s neuroprotective effects extend beyond amyloid pathways, suggesting supplementation could theoretically buffer both amyloid-dependent and amyloid-independent mechanisms of APOE4-related neurodegeneration.
Neuroinflammation amplification: APOE4 disrupts the microglia TREM2-APOE signaling axis and exacerbates neuroinflammation by activating the TLR4/NF-κB pathway. Lithium deficiency induces pro-inflammatory microglial activation, potentially creating a compounding inflammatory burden in APOE4 carriers that accelerates neurodegeneration.
Blood-brain barrier compromise: APOE4 compromises BBB integrity and activates the CypA-MMP9 pathway, facilitating infiltration of peripheral inflammatory cytokines and cerebrovascular pathology. This could theoretically affect brain lithium homeostasis, though this mechanism remains unexplored.
Distinct etiological pathways: Recent meta-analyses demonstrate that APOE ε4 defines distinct etiological pathways for dementia. See the work of Dr. Juan Fortea who I believe is a torch bearer for describing Apoe4 dementia as it’s own distinct typology! For example, diabetes specifically increases AD risk only in APOE ε4 noncarriers, while certain cardiovascular risk factors show stronger associations in carriers. This genotype-specific risk modification suggests that lithium deficiency might similarly interact differently with APOE4 versus non-APOE4 backgrounds.
Safety Considerations
Low-dose lithium appears relatively safe in older adults. Studies show it does not impair renal function at subtherapeutic doses over 2-4 years, though monitoring remains essential. Common side effects at common therapeutic doses (think, say, 900mg lithium carbonate, not 1mg lithium orotate) include thyroid dysfunction, weight gain, and polyuria, but these are less pronounced at lower doses. Recent 2025 data suggest adverse effects may emerge at lower serum levels than previously thought, reinforcing the need for careful monitoring even with low-dose supplementation. Even at low doses, it is wise to discuss supplementation with your health care provider. Certain medications, like Hydrochlorothiazide, may increase the serum lithium level in your body and thus, may require additional monitoring.
Clinical Implications and Future Directions
These findings suggest a potentially actionable insight: maintaining optimal brain lithium levels could represent an early, preventive strategy, particularly for genetically at-risk populations. However, several critical gaps remain:
No APOE-stratified lithium trials exist: Despite extensive research on both lithium in neurodegeneration and APOE4 in AD pathogenesis, no studies have examined lithium supplementation outcomes stratified by APOE genotype.
Optimal formulation unclear: While lithium orotate showed reduced amyloid binding in preclinical studies, clinical evidence comparing different lithium salts is limited and conflicting. The existing clinical trial evidence demonstrating cognitive benefits used lithium carbonate at subtherapeutic doses.
Dosing strategy undefined: The optimal lithium dosing for neuroprotection in APOE4 carriers—whether subtherapeutic (0.25-0.5 mEq/L) or higher—remains unknown.
Timing of intervention: Whether lithium supplementation is most effective in presymptomatic APOE4 carriers, those with MCI, or established AD requires investigation.
Conclusion
The discovery that lithium deficiency may be an early event in AD pathogenesis represents a paradigm shift in understanding this trace element’s role in brain health.For APOE4 carriers—who face substantially elevated AD risk through multiple converging pathways including impaired amyloid clearance, lipid metabolism disruption, neuroinflammation, and BBB dysfunction—lithium deficiency could represent an additional, modifiable risk factor.
Future clinical trials should prioritize APOE genotype stratification to determine whether lithium supplementation can meaningfully slow cognitive decline or delay AD onset in this genetically at-risk population. Given that APOE4 defines distinct etiological pathways for dementia, a precision medicine approach considering both genetic risk and lithium status may prove essential for optimizing preventive strategies.
For APOE4 carriers, this emerging evidence underscores that cognitive preservation may depend not only on traditional lifestyle interventions but also on maintaining optimal levels of trace elements like lithium that play foundational roles in brain resilience during aging.
Personally, I am taking 5mg of lithium orotate daily, mid-day, with my meal, and with my other supplements that I take as an APOE4 carrier, along with a daily dose of optimism. It’s truly a great time to be an Apoe4 carrier (and what choice do we have?!), as more and more research is coming out. Whether you choose to take lithium orotate is a personal choice that should be made in concert with your own healthcare provider, but for an asympomatic carrier such as myself, I am grateful that we have this elemental insight and I am not going to let it go to waste!


This is an unusually thoughtful synthesis, because it treats “lithium” not as a psychiatric drug headline, but as a trace element with plausible systems biology relevance to brain aging.
What I found most compelling is the reframing: endogenous lithium appears to be present in the brain, and in the Harvard/Rush datasets it’s reduced already at the MCI stage, while blood levels don’t necessarily track brain bioavailability. Mechanistically, the idea that amyloid can sequester lithium (turning plaques into a functional “sink”) provides a concrete bridge between the amyloid story and the long-standing puzzle of why pathology and symptoms don’t map 1:1. 
The downstream biology you highlight also fits what we see across neurodegeneration: lithium deficiency in models is associated with glial activation, synaptic and myelin-related injury signatures, and transcriptomic shifts overlapping with Alzheimer’s, suggesting a broad “network fragility” effect rather than a single-pathway tweak. 
Two clinician-facing nuances feel important:
1. This is not a green light for self-supplementation. Lithium has a narrow safety margin at pharmacologic doses, with renal/thyroid risks, drug interactions, and special concerns in older adults, so translating “endogenous depletion” into “replacement” needs properly designed human trials and careful formulation/dosing work. 
2. The biomarker implication is intriguing (brain-specific depletion, early change), but we’ll need clarity on what to measure (serum vs other compartments), how it behaves longitudinally, and how it performs against established risk markers. 
Overall: this is exactly the kind of hypothesis that can sharpen the field; less ideology (“amyloid vs not”) and more physiology (“what becomes unavailable, when, and to which cell types?”). If the lithium-bioavailability story holds up, it could open a prevention lane that’s both mechanistically grounded and clinically testable.
Excellent article. Curious, have you looked into whether dietary lithium from drinking water sources shows any meaningful correlation with cognitive outcomes in APOE4 carriers specifically?